|
Here's how you calculate the molar mass of H₂O by hand: So we multiply 2 by 1.008, then we multiply 1 by 15.999, and finally add up the two resulting numbers. 1 molecule of H₂O has 2 atoms of hydrogen and 1 atom of oxygen. Now, let me recall to you the atomic mass of lithium, sodium and potassium. The main thing I want to tell you is that he arranged these elements in the increasing order of their ATOMIC MASS. Oxygen has an average atomic mass of 15.999. Calcium (Ca), strontium (Sr) and Barium (Ba) in other triad and, Chlorine (Cl), bromine (Br) and iodine (I) in other triad. Hydrogen has an average atomic mass of 1.008. This is also the featured periodic table on the cover of our Periodic Table Journal available at Amazon. It includes element names, symbols, groups, atomic numbers, and atomic masses. It features our favorite color scheme of all the tables we’ve made. You can find the average atomic mass for any element on the periodic table of elements. This 118 element periodic table is a 1920×1080 HD wallpaper. In order to calculate molar mass by hand, you need to look up average atomic mass for each element in your compound. Therefore, 1 molecule of H₂O weighs 18.02 g/mol (g/mol is the unit used for molar mass). So 1 atom of hydrogen weighs 1.008 amu (a unit used to measure weight of atoms). This is the sum of all masses (2.016 + 15.999) in this case, which equals 18.02. However, if we have 6. The bottom of the table says (Molar Mass). According to the periodic table, the atomic mass of aluminum is 26.98 amu, copper is 63.55 amu, and carbon is 12.01 amu.Since 1 amu is only 1.674 × 10 24 g, these masses would be way too small to measure on ordinary laboratory equipment. There is only 1 atom of oxygen in one 1 molecule of water, so we multiply 1 by 15.999, which equals 15.999. These can be found in eye drops, mild antiseptics, washing powders and tile glazes. The most important compounds of boron are boric (or boracic) acid, borax (sodium borate) and boric oxide. It gives the flares a distinctive green colour. There are 2 atoms of hydrogen in 1 molecule of water, so we multiply 2 by 1.008, which equals 2.016. Amorphous boron is used as a rocket fuel igniter and in pyrotechnic flares. In both cases, it is the mass of 6.02 × 1023 molecules. For example, if we are trying calculate for ammonia (NH 3 ), then we. The atomic mass is equal to the atomic number which is listed below the element symbol. One mole of carbon dioxide molecules has a mass of 44.01g. Molar Mass of Ammonia NH 3 Step 1: The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. This is found by multiplying column two (Average Atomic Mass) by column three (Atoms). The molar mass of any compound is the mass in grams of one mole of that compound. Since H₂O has 2 atoms of hydrogen and 1 atom of oxygen, it says 2 for H and 1 for O.  
The third column is (Atoms), which shows the number of each atom found in the given molecule. The average atomic mass of hydrogen is 1.008, and the average atomic mass of oxygen is 15.999. The average atomic mass is found on the periodic table of elements as the bottom number. The average atomic mass is a weighted average of all of the isotopes of a given element. The second column says (Average Atomic Mass). In this case, it says H and O because H and O are the two elements found in the molecule H₂O, or water. The first column (Element) shows the symbols of the elements in the chemical given. Below this, you'll see a table which breaks the calculation down step-by-step. The molar mass shows up in the green box. 
One mole of a substance has the same mass in grams that one atom or molecule has in atomic mass units.Say you need to calculate the molar mass of H₂O. The numbers in the periodic table that we identified as the atomic masses of the atoms not only tell us the mass of one atom in u but also tell us the mass of 1 mol of atoms in grams. One mole of a substance has the same mass in grams that one atom or molecule has in atomic mass units. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of Na atoms has an approximate mass of 23 grams. Interactive periodic table of elements - your complete guide to the elements including definition, mass & names of each chemical in the periodic table. The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. To use the calculator, simply input the chemical formula of the substance and click the 'Calculate' button. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of H atoms has a mass of approximately 1 gram. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. 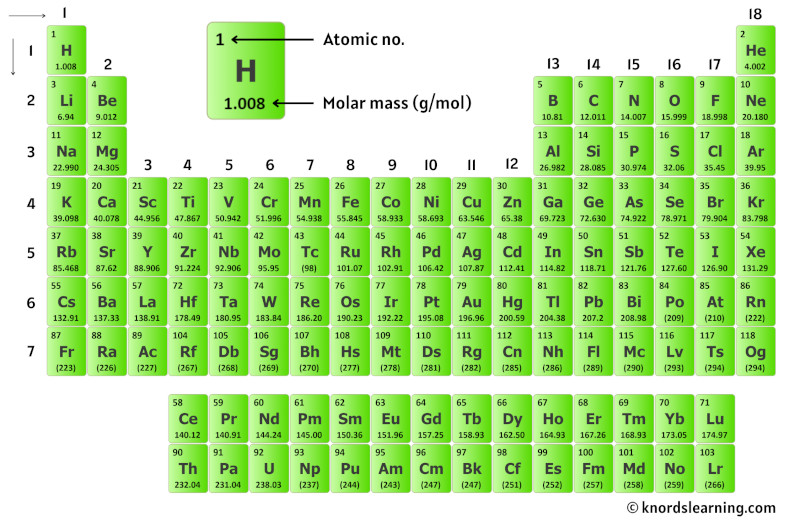
The number in a mole, Avogadro’s number, is related to the relative sizes of the atomic mass unit and gram mass units.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
